The Early Earth
The early Earth was a vision of hell, all scalding rock and choking fumes. Since then, its surface has cooled, continents have drifted, mountains have risen and eroded, and life has emerged, benign and green. Nearly all traces of the planet as it was have been wiped away. But from clues in the oldest rocks, deepest magmas, and even the cratered face of the moon, scientists have traced the planet's beginnings. As those early days have come into focus, so have the rare scenes, found today in some of Earth's harshest places that recall its ancient self.
Its birth pangs began some 4.6 billion years ago as rock and ice particles swirling around the young sun collided and merged, snowballing to produce ever-larger planetary building blocks. In violent pileups, they smashed together to create planets, including the infant Earth. In the turmoil, another body, as big as Mars, struck our planet with the energy of trillions of atomic bombs, enough to melt it all the way through. Most of the impactor was swallowed up in the bottomless magma ocean it created. But the collision also flung a small world's worth of vaporized rock into orbit. Debris quickly gathered itself into a ball, and since then Earth history has unfolded beneath the blank stare of the moon.
Early earth was a group of rocks in space pulled together over time by gravity. This superheated the earth, making it a large ball of molten rock. Over time, the earth eventually cooled down, and water was brought via asteroids that were melted. After this, primitive life began. Life then began to evolve over time, as new species were made and existing ones altered for the changing environment. As catastrophes stuck and changes occurred rapidly, evolution began to progress further. All of these changes eventually lead to the planet as we see today.

The Properties of Water
Polarity: Uneven distribution of charges across a molecule making one end positive (H) and the other negative (O).

Cohesion: The tendency for water molecules to form weak bonds and stick to each other.

Adhesion: the tendency of water to stick to other substances.

Capillary action: Adhesion allows water to stick to the sides of blood vessels or to the vascular tubes in plants. Both adhesion and cohesion allow water to move in one continuous column from a plant's roots to its leaves. This upward movement called capillary action.

Surface tension: a force that acts on the particles at the surface of a liquid. Surface tension is the tightness across the surface of water that is caused by polar molecules pulling on each other.

Specific heat: the amount of heat needed to increase the temperature of 1 kg of a substance by 1°C. Compared to other substances, water requires a lot of heat to increase its temperature.

Hydrogen bond: The positive hydrogen ends of one water molecule attract the negative oxygen ends of nearby water molecules causing them to stick together like weak magnets. This attraction causes water molecules to form temporary bonds that break easily. They are called hydrogen bonds.

1. Circle the letter of each sentence that is true about water's structure.
d. The chemical formula for water is H20.
2. One side of the water molecule has a positive charge, while the other side has a negative charge. What do the charges indicate about the molecule?
a. Water is a polar molecule.
c. Water is an ionic compound.
3. Bonds that form between water molecules are called
Hydrogen bonds.
4. True or False? Hydrogen bonds are strong and require a lot of energy to break.
False
5. The tendency for water molecules to stick to other water molecules is called Cohesion.
6. A force that acts on the particles of a liquid at the surface is called Surface Tension.
7. How does surface tension force the surface of water to curve? Gravity and surface tension forces on the water causing water to curve.
8. The tendency for water molecules to be attracted and stick to other substances is called Adhesion.
9. Circle the letter of each sentence that is true about water's surface tension.
a. It helps some insects "skate" across the surface of the water.
b. It refers to the tightness across the surface of the water.
c. It is caused by polar molecules repelling each other.
d. It causes raindrops to form round beads.
10. What is a curved surface in a graduated cylinder called? A meniscus.
11. Circle the letter of each sentence that is true about capillary action.
a. It explains how water moves against the force of gravity.
b. It is due to the attraction among molecules of water and surrounding materials.
d. It causes clothing to stay dry.
12. How does capillary action allow water to climb up the sides of a straw? Capillary action explains how water moves against the force of gravity, and is due to the attraction among molecules of water and surrounding materials. Capillary actions allow water to climb up the sides of a straw because the combined forces of cohesion and adhesion allow it to go against the forces of gravity.
13. A mixture that forms when one substance dissolves another is called a(n) Solution. The substance that does the dissolving is called a(n) Substance.
14. Why can water dissolve many substances? Water can dissolve many substances because it is polar. Water can dissolve substances like salt.
15. Circle the letter of each substance that dissolves in water.
a. salt
c. oxygen 16. A substance that hates, or “fears” water is called Hydrophobic
17. The amount of heat needed to increase the temperature of a certain mass of a substance by 1°C is its Specific Heat
18. True or False? Compared with other substances, water requires a lot of heat to increase its temperature. False
19. Circle the letter of each sentence that is true about water's specific heat.
a. It is due to the many attractions among water molecules
20. How does the high specific heat of water affect your body?
The high specific heat of water also helps your body to maintain a constant internal environment
Match the terms review
Match the terms review.
_____1. cohesion
_____2. capillary action
_____3. adhesion
_____4. polarity
_____5. hydrogen bond
_____6. surface tension
_____7. specific heat
a. tendency to stick to other substances
b. tightness caused by the pulling of water molecules on each other
c. the amount of heat needed to raise 1kg of a substance 1o
d. weak bonds formed between water molecules
e. tendency to stick to other water molecules
f. uneven distribution of charges in a molecule
g. tendency to climb up due to cohesion and adhesion properties of water
1. E
2. G
3. A
4. F
5. D
6. B
7. C
Salinity Lab

These were the materials we need for this lab: Salt, four clear water and the food coloring and medicine droppers

We put a couple of drops of red, blue and green in the beakers

Then we put 5 t-spoons in the all four of the beakers and mixed it.

Next ,we got the test tubes and medicine droppers and put salt water first, then green water, red water and blue water in the test tubes to make a rainbow

Next, we continued to put each of the colors and saw how it ended. So the colors made layers in the test tube and the reason why is by how much salt is in the water and the temperature.
Water Buoyancy Boats- Lab

These are the material we needed for the lab, aluminum foil and a basket of water

Then we cut out three 5 in x 5 in.

Next, we build three different boats so we can see how many pennies can be in each boat.

After we build the boats we place them in the water to see if they will float.

Lastly, we placed each boat pennies and see how many pennies will float until it will sink.
On the far left in took 3 pennies to sink, the one in the corner took 7 to sink and the middle took 3 to sink as well.
On the far left in took 3 pennies to sink, the one in the corner took 7 to sink and the middle took 3 to sink as well.
Water Density Presentation

DENSITY: noun ( pl. -ties) the degree of compactness of a substance : a reduction in bone density.
• Computing a measure of the amount of information on a storage medium (tape or disk). For magnetic tape it is the amount of information recorded per unit length of tape (bits per inch or millimeter); for a disk, a fixed number of bits per sector, sectors per track, and tracks per disk : chip density doubles every eighteen months | [as modifier, in combination ] a low-density 5.25-inch floppy disk | a drive capable of handling high-density 1.44 megabyte disks.
TEMPERATURE: (noun) the degree or intensity of heat present in a substance or object, esp. as expressed according to a comparative scale and shown by a thermometer or perceived by touch.
SALINITY: |səˈlinitē| (noun) containing or impregnated with salt : saline alluvial soils.
The ocean is a vast, extensive system that spans the entire globe. Within this system are currents that extend just as far as the ocean. These currents are driven by three major factors: density, temperature, and salinity. Density is how much weight water has compared to it's volume. Denser water sinks to the bottom. Temperature is a factor because cold water sinks and warmer water rises to the top. Salinity is the measure of how much salt is dissolved into water. Saltier water sinks to the bottom. All of these factors combine to create layers of differentiated values, which drive the ocean currents. These currents circulate the globe seeking equilibrium in water conditions, though since water is always being recycled and temperatures change over day and night, the currents always continue.
• Computing a measure of the amount of information on a storage medium (tape or disk). For magnetic tape it is the amount of information recorded per unit length of tape (bits per inch or millimeter); for a disk, a fixed number of bits per sector, sectors per track, and tracks per disk : chip density doubles every eighteen months | [as modifier, in combination ] a low-density 5.25-inch floppy disk | a drive capable of handling high-density 1.44 megabyte disks.
TEMPERATURE: (noun) the degree or intensity of heat present in a substance or object, esp. as expressed according to a comparative scale and shown by a thermometer or perceived by touch.
SALINITY: |səˈlinitē| (noun) containing or impregnated with salt : saline alluvial soils.
The ocean is a vast, extensive system that spans the entire globe. Within this system are currents that extend just as far as the ocean. These currents are driven by three major factors: density, temperature, and salinity. Density is how much weight water has compared to it's volume. Denser water sinks to the bottom. Temperature is a factor because cold water sinks and warmer water rises to the top. Salinity is the measure of how much salt is dissolved into water. Saltier water sinks to the bottom. All of these factors combine to create layers of differentiated values, which drive the ocean currents. These currents circulate the globe seeking equilibrium in water conditions, though since water is always being recycled and temperatures change over day and night, the currents always continue.
Marine Science: Bill Nye - Oceanography.
1. Heat from the sun makes the water in the ocean evaporate but the salt stays in the ocean because salt does not evaporate.
2. There is no salt in rain because the salt is not evaporated; therefore it cannot be in the rain.
3. Salty water is heavier than freshwater because there is dissolved matter in it making it denser.
4. Water helps keep England warmer because of warm water currents that come up from the south.
5. A current is a river of water flowing through water.
6. Thermohaline currents are driven by different values of heat and dissolved salt.
7. Data marker buoys are used to track ocean currents.
8. Fish in aquariums need currents so they can get food and water (with oxygen).
9. The Great Salt Lake and the Dead Sea are salty because they have no connection to the ocean to flush out their minerals.
10. The uneven heat in the water causes certain parts of the water to be less dense, so the lighter water and heavier water move around trying to find equilibrium.
11. The Atlantic Ocean has the most powerful current in the world.
12. Mars can't have oceans because Mars is too cold, so the water would freeze.
13. Many things depend on currents for transportation, food, and suitable climates, such as fish and algae.
2. There is no salt in rain because the salt is not evaporated; therefore it cannot be in the rain.
3. Salty water is heavier than freshwater because there is dissolved matter in it making it denser.
4. Water helps keep England warmer because of warm water currents that come up from the south.
5. A current is a river of water flowing through water.
6. Thermohaline currents are driven by different values of heat and dissolved salt.
7. Data marker buoys are used to track ocean currents.
8. Fish in aquariums need currents so they can get food and water (with oxygen).
9. The Great Salt Lake and the Dead Sea are salty because they have no connection to the ocean to flush out their minerals.
10. The uneven heat in the water causes certain parts of the water to be less dense, so the lighter water and heavier water move around trying to find equilibrium.
11. The Atlantic Ocean has the most powerful current in the world.
12. Mars can't have oceans because Mars is too cold, so the water would freeze.
13. Many things depend on currents for transportation, food, and suitable climates, such as fish and algae.
Tides
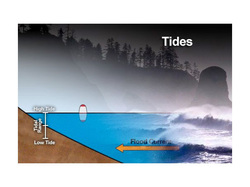
Tides are the regular, alternating rise and fall of sea level.
What Causes Tides?

Tides are caused by the gravitational pull of the moon and sun.
Where do tides occur?
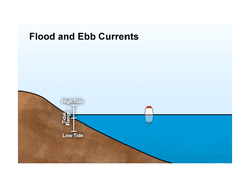
The changing of the tide is often rapid and dramatic. On a smaller scale, similar motions occur on large lakes, in the atmosphere, and even within the solid earth.
High and low tides
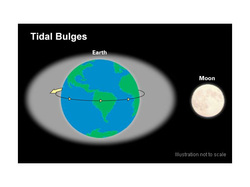
During a tidal cycle in the oceans, sea level rises, or floods, until it reaches its highest normal point, called high tide. Sea level drops, or ebbs, to its lowest point at low tide. The difference between high and low tide is the tidal range. These tides occur because forces generated by the gravitational pull of the sun and the moon, and by the rotation of the earth, tug seawater into two enormous bulges. One bulge is located on the edge of the planet closest to the moon. The other bulge forms on the opposite side of the planet.
Which object exerts the most forces on the tidal bulge? Why?
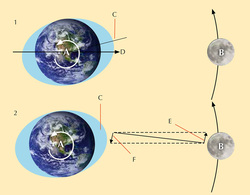
The sun is 27 million times more massive than the moon, but it is also almost 400 times farther away. As a result, the moon exerts more than twice as much tidal force on the oceans, so that a tidal bulge forms beneath and follows the moon. Solar gravity acts only to reinforce or diminish the moon’s pull. The other tidal bulge forms on the far side of the planet. As the earth rotates through space, the water on it tends to keep moving in a straight line. This tendency is able to tug the sea up into a bulge where the moon’s gravitational force is weakest. These bulges are the basis of Earth’s tides, but many other factors also influence the behavior of tides.
What happens when the sun and the moon align? What happens when they are perpendicular to each other?
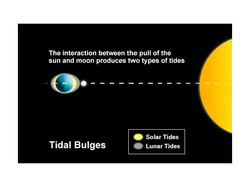
When the sun and moon align, their gravitational forces combine to produce the very highest and lowest tides, called spring tides. When the sun and moon are at right angles, they pull at the sea from different directions and moderate tides, with a smaller range, form. These are called neap tides.
What are some of the other factors that affect tides?

On a more local scale, the timing, size, and speed of tides varies with the shape of the coastline, seafloor topography, river discharge, and even wind and weather conditions.
What are the zones of the intertidal zone?
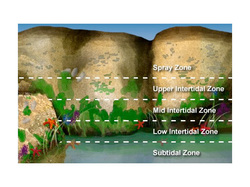
many areas have two, equal high tide – low tide cycles each day. In some regions though, the two cycles have different heights, and in still others, there is only one high and low tide each day. In all of these circumstances, the once or twice daily rise and fall of sea level poses enormous challenges and opportunities for the plants and animals that live in the dynamic space between high and low tides. This challenging habitat is known as the intertidal zone. It has four subdivisions—the spray zone, and the upper, middle, and lower intertidal zones:
The spray zone is wetted by the splash of breaking waves, but rarely submerged. This zone supports very limited life.
The high intertidal zone is underwater only during high tide.
The middle intertidal zone is mostly submerged except for brief periods once or twice a day during low tide.
The lower intertidal zone is only exposed to air during the lowest spring tides.
The spray zone is wetted by the splash of breaking waves, but rarely submerged. This zone supports very limited life.
The high intertidal zone is underwater only during high tide.
The middle intertidal zone is mostly submerged except for brief periods once or twice a day during low tide.
The lower intertidal zone is only exposed to air during the lowest spring tides.
What are some of the challenges for the critters of the intertidal zone?

In order to survive, they must be able to avoid drying out, endure temperature extremes, withstand intense sunlight, and survive a wide range of salinities. They also must escape predation by land animals during their long exposure.
How do the intertidal organism COPE with the harsh environment?
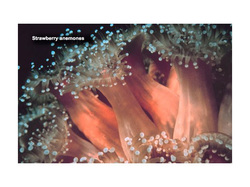
Intertidal organisms cope with these harsh conditions in several ways. Seaweeds and other plants have tough leathery leaves that resist water loss. They grow in clumps that collapse upon one another when stranded so that only the top layer is exposed to sunlight. Soft bodied animals, like anemones, crowd together to reduce their surface area. Others scrunch up their bodies and secrete a protective coating of mucous when out of water. Shelled animals, like snails and clams, close up tightly to conserve moisture and repel predators. Mobile animals retreat with the falling tide, burrow into sandy or rocky shelters, or hide beneath overhanging rocks and amid plants.
What are some of the adaptations that help organisms cope with wave action?

Organisms have developed a variety of adaptations to survive wave and tidal action. Sea stars have a thick skin and strong skeleton that can handle wave energy. They also have powerful arms lined with suction like tube feet to hold on tight in moving water. Sea slugs and other snails have a large foot that grips tightly to surfaces as they move or rest. Many other invertebrates, such as clams, and mussels have very thick shells, and cement themselves to rocks and one another for strength against the waves. Lower intertidal plants sway easily with the waves and currents because of firmly anchored roots and flexible stems. Fish that frequent the intertidal zone tend to have sturdy bodies and strong muscles.
